Resetting the curve – circadian rhythm's effects on the eye
Circadian rhythms (CR) shape almost every aspect of physiology, from metabolism and hormone release to vascular tone and mood. For eyecare professionals, understanding these daily cycles is increasingly relevant: the eye is both a detector and a regulator of circadian timing. Disrupted rhythm has been linked not only to systemic conditions, such as diabetes and hypertension, but also to ocular diseases where light, sleep and stress biology intersect. Here, I outline key circadian mechanisms and explore emerging links to glaucoma and central serous chorioretinopathy (CSCR).
Circadian rhythms are internal, self-sustaining cycles that regulate physiology over roughly 24 hours. Unlike diurnal rhythms, which respond directly to environmental change, CRs persist even in the absence of external cues. This autonomy arises from molecular clocks operating within almost all cells, coordinated by the suprachiasmatic nucleus (SCN) in the anterior hypothalamus, which synchronises peripheral clocks across organs such as the liver, heart and retina.
At the cellular level, CRs stem from transcription-translation feedback loops. CLOCK and BMAL1 proteins activate PER (period) and CRY (cryptochrome) genes, which accumulate and inhibit CLOCK/BMAL1 gene activity, completing a 24-hour cycle. Secondary loops, involving REV-ERB and ROR proteins, modulate BMAL1 expression to stabilise phase timing, while post-translational changes fine-tune precision (Fig 1).
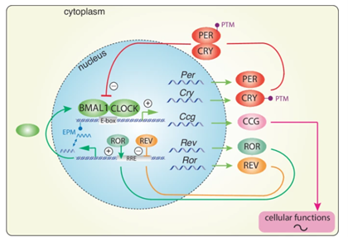
Fig 1. CCG’s impact on cellular functions1
These cellular clocks regulate a wide range of processes – metabolism, hormone release, vascular tone, immune function – producing predictable daily rhythms. In the liver, for instance, the genes promoting glucose uptake dominate during the day, while those driving gluconeogenesis take over at night, maintaining energy balance during fasting.
Because their natural cycle is close to, but not exactly, 24 hours, circadian rhythms must be continually re-synchronised. This is achieved through zeitgebers (‘time givers’) – environmental and behavioural cues that reset the clock. Light is the most powerful, followed by factors such as meal timing, activity, social interaction, temperature and stress (Table 1).
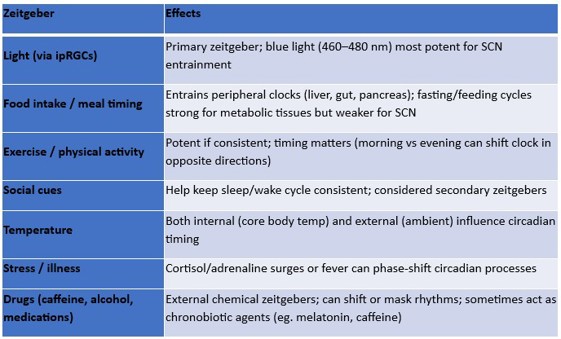
Table 1. Summary of zeitgebers and their effects
For eyecare professionals, the visual system’s role in this synchronisation is central. While rods and cones transmit image-forming signals to the visual cortex, circadian entrainment relies on a separate, non-image-forming pathway. A specialised subset of retinal ganglion cells – the intrinsically photosensitive retinal ganglion cells (ipRGCs) – contain melanopsin, a photopigment most sensitive to blue light (460–480nm). These cells project via the retinohypothalamic tract to the SCN, aligning circadian rhythms with environmental light and influencing sleep-wake timing, hormone secretion and mood (Fig 2).
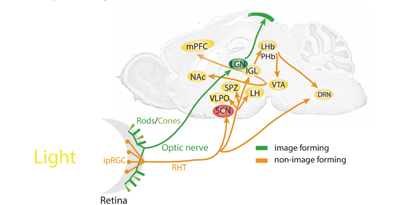
Fig 2. Light’s visual pathways1
Within the SCN, ipRGC activation triggers the release of glutamate and pituitary adenylyl cyclase-activating peptide (PACAP). Glutamate acts on NMDA receptors, leading to calcium influx and activation of PER and CRY transcription. PACAP, which is co-released with glutamate, amplifies this signal through cAMP and CREB-mediated pathways, promoting PER1 and PER2 expression.
The result is a precise light-dependent phase adjustment: morning light advances the clock, while evening light delays it. PACAP becomes particularly important during major shifts, such as jet-lag or night-shift adaptation, when the circadian system must rapidly recalibrate (Table 2).
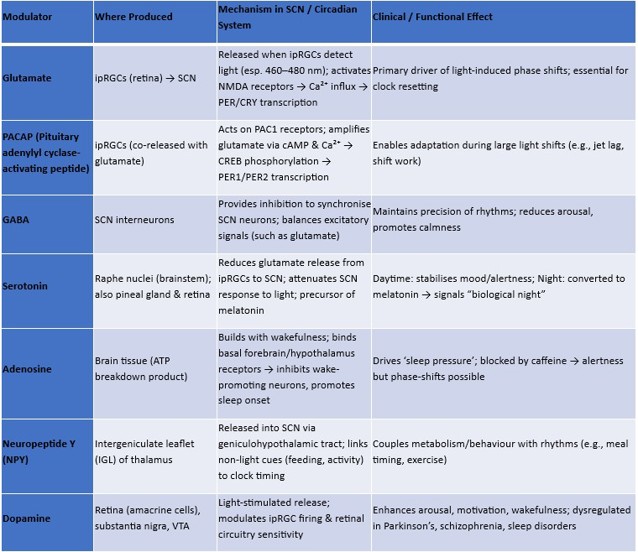
Table 2. Modulators of circadian sensitivity
Disruption, disease and chronotherapy
The SCN doesn’t operate in isolation; its outputs ripple across the body, producing rhythms in melatonin and cortisol secretion, body temperature, cardiovascular tone, alertness and immune function. During daylight, ipRGC activity suppresses sympathetic outflow to the pineal gland, preventing melatonin synthesis. At night, when ipRGCs fall quiet, the SCN lifts this inhibition, allowing noradrenaline release to trigger melatonin production. This nocturnal rise promotes sleep onset and synchronises peripheral clocks throughout the body.
Clinical pearl – CR extends far beyond sleep. Core body temperature falls before sleep and peaks in the evening; sympathetic tone rises in the morning, while parasympathetic activity dominates at night; and hormones, such as growth hormone, prolactin and testosterone, follow predictable daily patterns. Even cognitive performance, mood and immune responsiveness oscillate with these rhythms.
Humans, however, are unusual in their ability to override their biological clock. Behavioural factors, such as night-shift work, transmeridian travel and irregular light exposure, can desynchronise internal timing. Pharmacological and environmental factors add further complexity. Caffeine and alcohol can mask circadian signals, while artificial lighting – particularly blue-enriched LED and screen light – can shift or flatten rhythmic output.
That syncing feeling
Chronic circadian disruption is now recognised as a contributing factor to multiple systemic disorders. Misalignment between central and peripheral clocks is linked to impaired glucose metabolism, hypertension, depression and poor sleep quality. In ocular tissues, circadian mechanisms govern intraocular pressure, aqueous humour dynamics, retinal metabolism and vascular tone. When these local rhythms fall out of step with systemic ones, it can result in disease progression or treatment resistance.
This connection has inspired interest in chronotherapy – timing medical interventions to integrate with biological rhythms to enhance their effectiveness and reduce harm. Cardiovascular events, such as myocardial infarction and stroke, peak in the early morning, corresponding to surges in blood pressure and platelet activity. Consequently, antihypertensive therapy timing has become an area of active research. In ophthalmology, excessive nocturnal blood-pressure dipping can reduce ocular perfusion pressure, potentially accelerating glaucoma progression. The clinical lesson is not that medication should universally shift to night-time dosing, but that timing should reflect the individual’s risk profile and rhythm.
Early chronotherapy trials often targeted hormones with clear circadian profiles, such as melatonin, orexin (via agents such as modafinil), or dopamine pathways, in myopia control. Yet manipulating a single hormone seldom restores the broader circadian network and can introduce side effects. Melatonin, for instance, remains popular but has recently been withdrawn from some Australian online retailers following reports of paediatric overdoses.
Part of the challenge lies in methodology. Drug studies lend themselves to controlled conditions, but zeitgebers – such as light, feeding, or activity – are complex, context-dependent and harder to isolate. A tightly controlled laboratory study may reveal a mechanism but lack ecological validity for real-world behaviour. Increasingly, researchers recognise that ‘messier’ study designs involving individuals in their natural environments can produce more clinically meaningful insights.
This shift has led to a focus on behavioural and environmental chronotherapy. Light therapy is now used to manage cancer-related fatigue and neurodegenerative disorders, time-restricted feeding is being trialled for obesity and shift-work adaptation, and scheduled activity can reinforce circadian stability. In Australia, the Consumers Health Forum and the Royal Australian College of General Practitioners, supported by the NHMRC Partnership Centre, have begun developing frameworks for ‘social prescribing’ – embedding such rhythm-aligned strategies into primary and preventive healthcare.
Spotlight: melatonin and glaucoma
Melatonin is best known as the ‘sleep hormone’, but its physiological influence extends well beyond the pineal gland. Under control of the SCN, melatonin secretion rises at night and falls sharply in response to light exposure through the melanopsin-containing ipRGC pathway. Within the eye, it acts locally as well as systemically. MT₁ and MT₂ receptors are expressed in the ciliary body, trabecular meshwork and retina, where activation influences aqueous dynamics and provides antioxidant, anti-inflammatory and neuroprotective effects.
Melatonin levels follow a circadian rhythm – low during daylight, rising steeply in the evening and peaking overnight. Disruption of this pattern has been reported in glaucoma, particularly in advanced disease2-4. Of special relevance are the ipRGCs themselves. These ganglion cells mediate circadian photoentrainment and contribute to retinal signalling, but they are selectively lost in glaucoma – up to 70% of them in severe cases5. Their degeneration may therefore not only impair visual processing but also reduce systemic light input to the SCN, subtly altering sleep, mood and metabolic regulation6-10.
Understanding melatonin research requires attention to how it is measured. Serum assays capture total melatonin, most of which is protein-bound and biologically inactive. Salivary testing reflects the free, active fraction, while urinary 6-sulfatoxymelatonin provides a cumulative overnight index. The timing of sampling adds further variability: some studies use fixed clock times, others use dim-light melatonin onset (DLMO) – the rise in melatonin under controlled low-light conditions. DLMO remains the most reliable marker of circadian phase, but differences in assay type and sampling protocol explain much of the inconsistency across the literature.
In animal models, exogenous melatonin consistently lowers intraocular pressure (IOP) through MT₁ and MT₂ receptor activity in the ciliary body and trabecular meshwork2. Small human trials generally support this trend, reporting modest nocturnal IOP reductions and possible additive effects alongside standard glaucoma therapies11-14. Melatonin analogues, such as agomelatine and ramelteon, show similar pharmacological activity, though clinical data remain limited and short-term.
Beyond pressure control, melatonin’s potential neuroprotective effects have drawn growing interest. Experimental work suggests it can reduce oxidative stress, dampen excitotoxicity and limit apoptosis within retinal ganglion cells15-19. These findings are especially relevant given the overlap between ipRGC loss and systemic circadian disturbance in glaucoma. It’s plausible that disruption of light-sensing pathways contributes to subtle changes in sleep or mood regulation, though the extent of this relationship in clinical populations remains to be clarified.
At present, melatonin’s role in glaucoma should be viewed as exploratory rather than established. It may eventually inform adjunctive therapies or serve as a biomarker of circadian involvement, but further longitudinal and mechanistic studies are needed. What is clear is that circadian physiology is embedded in ocular biology and understanding these interactions could refine how we think about both disease mechanisms and patient wellbeing.
Spotlight: cortisol and central serous chorioretinopathy
Cortisol, the body’s main endogenous steroid hormone, mirrors many of the physiological effects of prescribed corticosteroids. It regulates inflammation, vascular tone and energy metabolism through the hypothalamic-pituitary-adrenal (HPA) axis: corticotropin-releasing hormone (CRH) from the hypothalamus stimulates pituitary secretion of adrenocorticotropic hormone (ACTH), which in turn drives adrenal cortisol release.
Under normal conditions, cortisol levels rise sharply on waking – the cortisol awakening response (CAR) – and decline gradually to a nocturnal low. This rhythm, governed by the SCN, is highly sensitive to sleep quality, stress and illness. Even short-term disruption can flatten or delay the curve, reducing the amplitude between morning and night values. For accurate assessment, multi-point salivary or urinary sampling over several days is preferred, as single serum draws often miss these fluctuations (Fig 3).
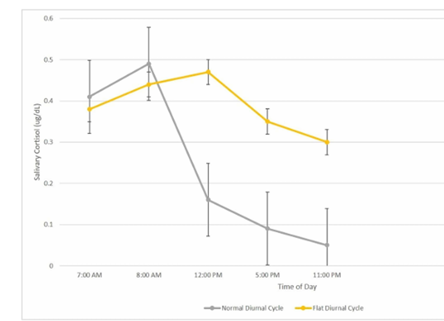
Fig 3. Normal diurnal cortisol output shows a sharp morning rise and gradual decline to a low point at night. In CSCR, this rhythm is often flatter, with a reduced morning peak and relatively higher evening levels. Credit: Speer KE, et al
Cortisol circulates mainly bound to carrier proteins, with only a small free fraction that is physiologically active. This unbound cortisol interacts with two receptor types: mineralocorticoid receptors (MRs), which respond to basal levels and maintain circadian tone, and glucocorticoid receptors (GRs), which activate during peaks or stress to modulate metabolism and immune function.
Links between cortisol and central serous chorioretinopathy (CSCR) have long been recognised, though the precise mechanism remains under study. Early work reported elevated urinary free cortisol21 and exaggerated CARs in CSCR22, suggesting heightened HPA activity. Later studies using more detailed diurnal profiling described a flatter rhythm, with reduced morning peaks and relatively higher evening levels. These blunted patterns correlated with greater choroidal thickness, vascularity index and flow-signal voids on OCT angiography – findings consistent with dysregulated vascular tone and permeability23.
Sleep and stress-related factors appear to compound this relationship. Shift-work exposure and obstructive sleep apnoea – both associated with altered cortisol dynamics – are more common among CSCR patients, while restoration of normal rhythm with continuous positive airway pressure (CPAP) has been linked to structural recovery in small studies24,25. Neurotransmitter systems may also contribute: reduced serotonin, a melatonin precursor that influences HPA regulation, has been associated with multifocal leakage and increased recurrence26.
Whether absolute cortisol excess or rhythmic distortion is the key driver is not yet clear. Differences in assay type, sampling window and population characteristics complicate comparison across studies. What does emerge is a consistent picture of circadian imbalance – one that aligns with CSCR’s known links to stress physiology and steroid sensitivity.
For clinicians, this highlights the potential value of interventions that support circadian stability: structured sleep, light exposure, exercise and stress management. Pilot studies using oral melatonin in non-resolving CSCR have hinted at benefit, though findings remain preliminary27. As with glaucoma, these insights suggest that understanding a patient’s broader physiological rhythms may one day complement our traditional disease models – not by replacing them, but by expanding how we think about systemic and ocular interaction.
Conclusion
Circadian physiology is not just background biology but an organising principle for health. The eye plays a central role in this system, translating light into neural and hormonal signals that synchronise the body’s internal clocks. For optometrists, recognising circadian influences offers new insight into familiar conditions – from intraocular pressure fluctuations in glaucoma to stress-related vascular changes in CSCR. As research continues, integrating circadian awareness into patient care may help us better understand not only when disease occurs, but why timing itself matters.
References
- Stewart D, Albrecht U. Beyond vision: effects of light on the circadian clock and mood-related behaviours. npj Biological Timing and Sleep. 2025 Mar 13;2(1):12.
- Martínez-Águila A, Martín-Gil A, Carpena-Torres C, Pastrana C, Carracedo G. Influence of circadian rhythm in the eye: significance of melatonin in glaucoma. Biomolecules. 2021 Feb 24;11(3):340
- Pintor J, Martin L, Pelaez T, Hoyle CH, Peral A. Involvement of melatonin MT3 receptors in the regulation of intraocular pressure in rabbits. European journal of pharmacology. 2001 Mar 30;416(3):251-4.
- Belforte NA, Moreno MC, De Zavalía N, Sande PH, Chianelli MS, Keller Sarmiento MI, Rosenstein RE. Melatonin: a novel neuroprotectant for the treatment of glaucoma. Journal of pineal research. 2010 May;48(4):353-64.
- Drouyer E, Dkhissi-Benyahya O, Chiquet C, WoldeMussie E, Ruiz G, Wheeler LA, Denis P, Cooper HM. Glaucoma alters the circadian timing system. PLoS One. 2008 Dec 12;3(12):e3931.
- Ma XP, Shen MY, Shen GL, Qi QR, Sun XH. Melatonin concentrations in serum of primary glaucoma patients. International Journal of Ophthalmology. 2018 Aug 18;11(8):1337.
- Gubin DG, Malishevskaya ТN, Astakhov YS, Astakhov SY, Cornelissen G, Kuznetsov VA, Weinert D. Progressive retinal ganglion cell loss in primary open-angle glaucoma is associated with temperature circadian rhythm phase delay and compromised sleep. Chronobiology international. 2019 Apr 3;36(4):564-77.
- Wang H, Zhang Y, Ding J, Wang N. Changes in the circadian rhythm in patients with primary glaucoma. PLoS One. 2013 Apr 29;8(4):e62841.
- Guo ZZ, Jiang SM, Zeng LP, Tang L, Li N, Xu ZP, Wei X. ipRGCs: possible causation accounts for the higher prevalence of sleep disorders in glaucoma patients. International journal of ophthalmology. 2017 Jul 18;10(7):1163.
- Zhang Y, Huang Y, Guo L, Zhang Y, Zhao M, Xue F, Tan C, Huang J, Chen D. Melatonin alleviates pyroptosis of retinal neurons following acute intraocular hypertension. CNS & Neurological Disorders-Drug Targets-CNS & Neurological Disorders). 2021 Mar 1;20(3):285-97.
- Pescosolido N, Gatto V, Stefanucci A, Rusciano D. Oral treatment with the melatonin agonist agomelatine lowers the intraocular pressure of glaucoma patients. Ophthalmic and physiological optics. 2015 Mar;35(2):201-5.
- Ismail SA, Mowafi HA. Melatonin provides anxiolysis, enhances analgesia, decreases intraocular pressure, and promotes better operating conditions during cataract surgery under topical anesthesia. Anesthesia & Analgesia. 2009 Apr 1;108(4):1146-51.
- Carracedo-Rodríguez G, Martínez-Águila A, Rodriguez-Pomar C, Bodas-Romero J, Sanchez-Naves J, Pintor J. Effect of nutritional supplement based on melatonin on the intraocular pressure in normotensive subjects. International ophthalmology. 2020 Feb;40(2):419-22.
- Samples JR, Krause G, Lewy AJ. Effect of melatonin on intraocular pressure. Current eye research. 1988 Jan 1;7(7):649-53.
- Kaur C, Sivakumar V, Yong Z, Lu J, Foulds WS, Ling EA. Blood–retinal barrier disruption and ultrastructural changes in the hypoxic retina in adult rats: the beneficial effect of melatonin administration. The Journal of Pathology: A Journal of the Pathological Society of Great Britain and Ireland. 2007 Aug;212(4):429-39.
- Diéguez HH, González Fleitas MF, Aranda ML, Calanni JS, Keller Sarmiento MI, Chianelli MS, Alaimo A, Sande PH, Romeo HE, Rosenstein RE, Dorfman D. Melatonin protects the retina from experimental nonexudative age‐related macular degeneration in mice. Journal of Pineal Research. 2020 May;68(4):e12643.
- Liang FQ, Green L, Wang C, Alssadi R, Godley BF. Melatonin protects human retinal pigment epithelial (RPE) cells against oxidative stress. Experimental eye research. 2004 Jun 1;78(6):1069-75.
- Rosen RB, Hu DN, Chen M, McCormick SA, Walsh J, Roberts JE. Effects of melatonin and its receptor antagonist on retinal pigment epithelial cells against hydrogen peroxide damage. Molecular Vision. 2012 Jun 20;18:1640.
- Chang CC, Huang TY, Chen HY, Huang TC, Lin LC, Chang YJ, Hsia SM. Protective effect of melatonin against oxidative stress‐induced apoptosis and enhanced autophagy in human retinal pigment epithelium cells. Oxidative Medicine and Cellular Longevity. 2018;2018(1):9015765.
- Speer KE, Semple S, Naumovski N, D'Cunha NM, McKune AJ. HPA axis function and diurnal cortisol in post-traumatic stress disorder: A systematic review. Neurobiology of stress. 2019 Nov 1;11:100180.
- Haimovici R, Rumelt S, Melby J. Endocrine abnormalities in patients with central serous chorioretinopathy. Ophthalmology. 2003 Apr 1;110(4):698-703
- Scarinci F, Patacchioli FR, Ghiciuc CM, Pasquali V, Bercea RM, Cozma S, Parravano M. Psychological Profile and distinct salivary cortisol Awake Response (CAR) in two different study populations with obstructive sleep apnea (OSA) and Central Serous Chorioretinopathy (CSC). Journal of Clinical Medicine. 2020 Aug 3;9(8):2490.
- Scarinci F, Patacchioli FR, Costanzo E, Parravano M. Cortisol awake response imbalance as an indicator of acute central serous chorioretinopathy: Relationship with choriocapillaris and choroidal features. Frontiers in Medicine. 2022 Nov 30;9:1030352.
- Bousquet E, Dhundass M, Lehmann M, Rothschild PR, Bayon V, Leger D, Bergin C, Dirani A, Beydoun T, Behar-Cohen F. Shift work: a risk factor for central serous chorioretinopathy. American Journal of Ophthalmology. 2016 May 1;165:23-8.
- Leveque TK, Yu L, Musch DC, Chervin RD, Zacks DN. Central serous chorioretinopathy and risk for obstructive sleep apnea. Sleep Breath. 2007 Dec;11(4):253-7. doi: 10.1007/s11325-007-0112-3. PMID: 17457629.
- Kimura T, Araki T, Komuku Y, Iwami H, Gomi F. Central serous chorioretinopathy and blood serotonin concentrations. Journal of Clinical Medicine. 2021 Feb 3;10(4):558.
- Gramajo AL, Marquez GE, Torres VE, Juarez CP, Rosenstein RE, Luna JD. Therapeutic benefit of melatonin in refractory central serous chorioretinopathy. Eye. 2015 Aug;29(8):1036-45.

Layal Naji is an Australian optometrist and lecturer in optometry at the University of Canberra, currently completing a PhD on real-world outcomes of emerging therapies for inherited retinal disease. She is a Fellow of the Higher Education Academy (FHEA) and a regular contributor to NZ Optics.